The fascinating story of the life-cycle of steel begins with iron, which is found in the Earth’s crust only in the form of an ore, usually an iron oxide, such as magnetite, hematite etc. Iron is extracted from iron ore by removing the oxygen and combining the ore with a preferred chemical partner such as carbon. Before the introduction of the Bessemer process in the mid 19th century steel was expensive and was only used where no cheaper alternative existed, for example in the cutting edge of knives, razors, swords, and other items where a hard, sharp edge was needed. It was also used for springs, including those used in clocks and watches.
Further refinements in the process, such as basic oxygen steelmaking, lowered the cost of production while increasing the quality of the metal. Today, steel is one of the most common materials in the world, with more than 25 million tonnes of products produced annually. The steel industry is responsible for employing around 2 million people worldwide, and providing we always remember to recycle it, this material will help to keep these people in jobs for years to come.
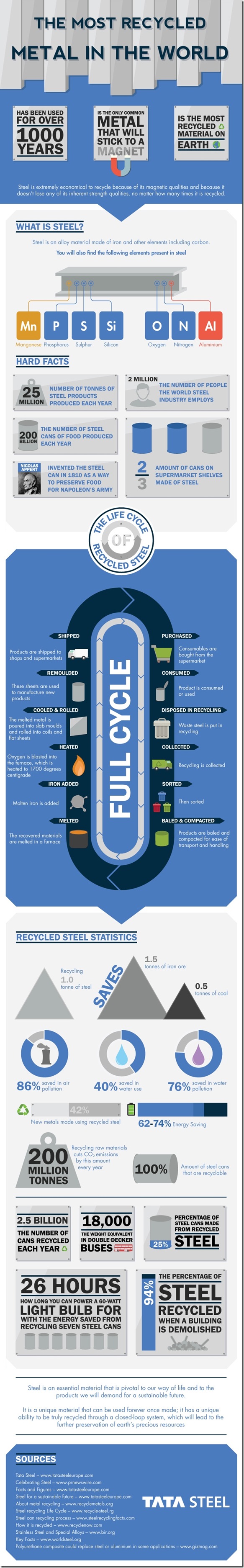
Steel is extremely economical to recycle because of its magnetic qualities and because it does not lose any of its inherent strength qualities not matter how many times it is recycled. 1.5 tonnes of ore and 0.5 tonnes of coal are saved by recycling 1 tonne of steel and recycling also saves 86% in air pollution, 76% in water pollution and 40% in water used. Once the steel has been made into a product for either business or public consumers it begins the start of its fascinating recycling circle. Our fun infographic shows the many stages of this journey, as well as providing more facts and figures about this amazing material.